SOLTOSTEN
- Without prescription
- Download instruction
- Share this:
- ABOUT PRODUCT
- INSTRUCTIONS
COMPOSITION AND DOSAGE FORM
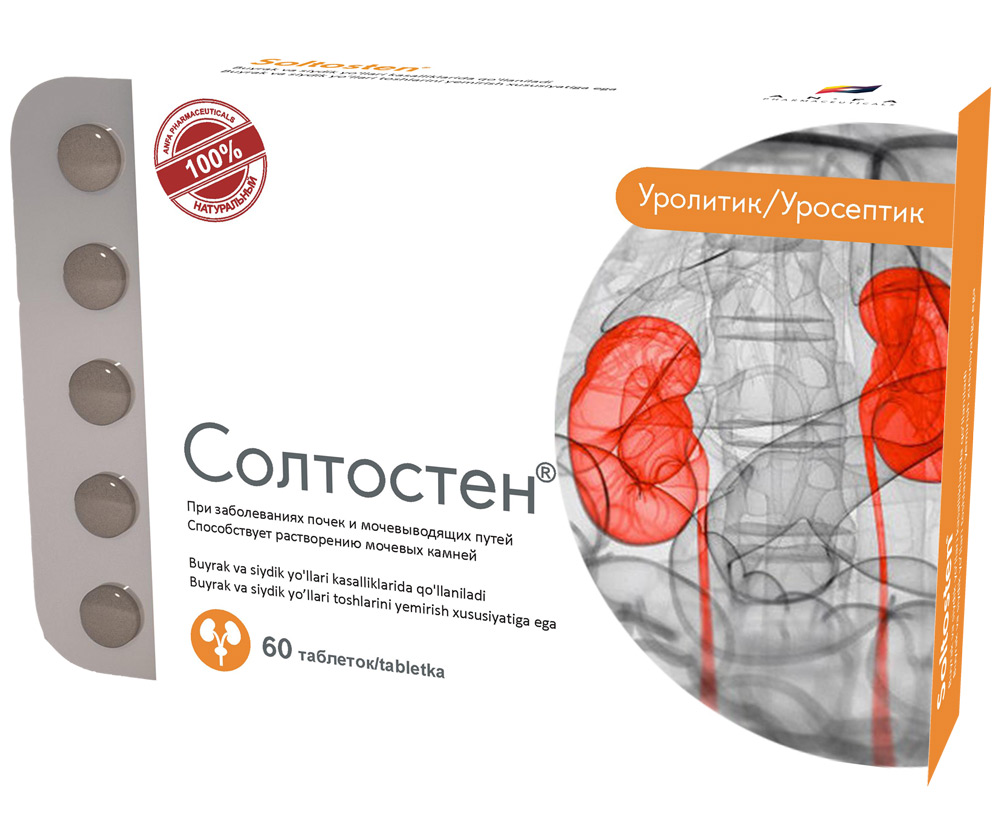
Round tablets of herbs, with individual odor, biconvex, light – brown, speckled tablets. 60 pieces 0.5 g each. Polyethylene vials with a protective seal or packed in blisters. (1) – cardboard pack with protective holographic label, (2) – instruction for use.
Composition:
Yellow Marsh Saxifrage (Saxifraga hirculus L.)
Common Madder (Rubia tinctorum L.)
Common Horsetail (Equisetum arvense L..)
Indian Kidney Tea (Folium Orthosíphonis L.)
Clusterberry Leaf (Vaccinium vitis-idaea L.)
Mountain Knotgrass (Aerva lanata)
Prostrate Knot Grass (Polygonum aviculare L.)
Common Barberry (Berberis vulgaris L.)
EFFECT OF THE MEDICINE
- The medicine has antispasmodic, diuretic and anti-inflammatory effect.
- Promotes the discharge of stones of various nature in nephrolithiasis, normalizes salt metabolism.
SIDE EFFECTS
Usually, there are no side effects, in the majority of cases, the medicine is well tolerated.
In the case of individual sensitivity to one or more components of the medicine, there may be allergic reactions, dyspeptic disorders of the gastrointestinal tract.
Use of the medicine should be discontinued when hypersensitivity reactions occur.
INTERACTION WITH OTHER MEDICATION
When prescribing with other medication adverse effects were not revealed.
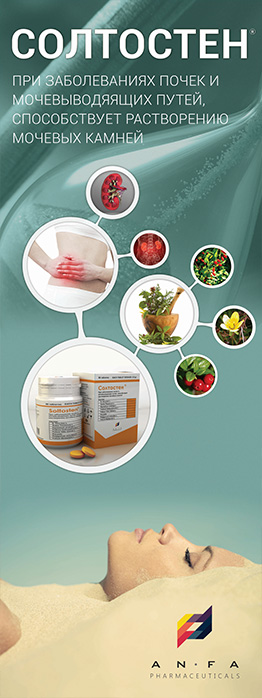
INDICATIONS
- Chronic inflammatory diseases of the kidney and urinary tract (pyelonephritis, pyelitis, cystitis, urethritis, prostatitis).
- Urolithiasis.
- Cholelithiasis, cholecystitis, cholangitis.
- Urine acid diathesis.
- Osteochondrosis, gout.
- Moreover, the medicine is prescribed in the postoperative period after removal of kidney stones or their spontaneous discharge.
CONTRAINDICATIONS
Hypersensitivity to one or more components of the medicine.
DOSAGE
The duration of treatment and the dose are determined by the attending physician for each patient individually.
CHILDREN 3 YEARS OF AGE AND ABOVE
ADULTS
1 tablet 2 times a day 5-10 minutes before the meal.
2 tablets 3 times a day 5-10 minutes before the meal.
The course of treatment in chronic diseases is usually 20 – 30 days.
If necessary the treatment course can be repeated.
OVERDOSE
The cases of SOLTOSTEN overdose were not reported.
STORAGE CONDITIONS AND EXPIRATION DATE
The medicine should be stored out of reach of children, protected from light, at a temperature from + 2° C to + 25°C.
Expiration date: 2 years.
The information presented on the site should not be used for self-diagnosis and treatment and can not serve as a substitute for a doctor's internal consultation.