GORLOZIN
- Without prescription
- Download instruction
- Share this:
- ABOUT PRODUCT
- INSTRUCTIONS
COMPOSITION AND PACKING
Syrup of herbal extracts, brown viscous liquid with individual odor (may have light sediment and clouding) in the original vials of 150 ml with a protective seal – (1) cardboard packs with protective branded holographic label; (2) Spoon – dispenser; (3) instruction for use.
Composition:
Liquorice Extract (Glycyrrhiza glabra)
Ginger Extract (Zingiber officinale)
Marigold Extract (Calendula officinalis)
Turmeric Extract (Curcuma longa)
Chamomile Extract (Matricaria chamomilla)
Aloe Extract (Aloe arborescens)
Piper Cubeba Extract (Piper cubeba L.)
EFFECT OF THE MEDICINE
– Shows fungicidal and virostatic action;
– Has anti-inflammatory, antiseptic and antibacterial effect;
– Increases immunity;
– Liquorice and Calendula reduce pain, promotes relaxation, relieves inflammation, covers, contributes to the removal of phlegm and gives an antiseptic effect;
– Chamomile relieves inflammation of the throat and tonsils;
– Turmeric relieves inflammation, destroys the germs;
– Ginger Officinale relieves inflammation;
– Piper Cubeba relieves inflammation, promotes tissue restoration;
– Aloe relieves inflammation and helps to restore tissue.
SIDE EFFECTS
There may be rarely allergic hypersensitivity to separate ingredients of the preparation. Use of the medicine should be discontinued when hypersensitivity reactions occur.
SPECIAL INSTRUCTIONS
Before use, carefully shake the vial. Keep the vial in a cool place after opening it. Do not heat and do not freeze.
INTERACTION WITH OTHER MEDICATION
When prescribing with other medication adverse effects were not revealed.
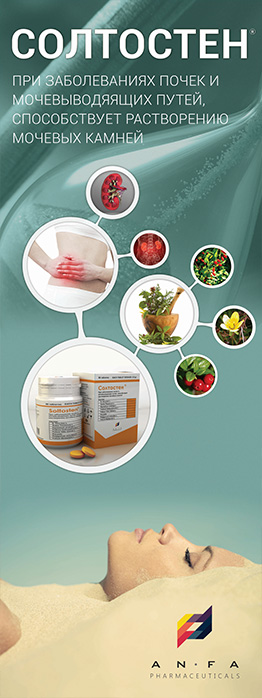
INDICATIONS
– Comprehensive Elimination of chronic and acute tonsillitis (for the oral mucosa treatment in patients with infectious and inflammatory diseases of the mouth);
– Acute laryngitis;
– Unspecified Acute tonsillitis (agranulocytic angina);
– Acute upper respiratory infections of multiple and unspecified sites;
– Chronic, atrophic pharyngitis
CONTRAINDICATIONS
Individual intolerance to certain ingredients of the medicine.
DOSAGE
The duration of treatment and the dose are determined by the attending physician for each patient individually.
Per os::
CHILDREN BETWEEN 1 AND 6 YEARS OF AGE
CHILDREN BETWEEN 7 AND 12 YEARS OF AGE
ADULTS AND CHILDREN ABOVE 12 YEARS OF AGE
1.0-2.0 ml 3 times a day
2.0-2.5 ml 3 times a day
2.5-5.0 ml 3 times a day
The medicine should be taken at least 15 days to achieve a therapeutic effect.
External use: Gargle at: Laryngitis and Tonsillitis: gargle with the solution (diluted with purified water 1: 6) 6-10 times a day
HOW TO GARGLE:
– When gargle it is necessary to tilt the head back and stick out the tongue forward as much as possible so the solution can pass into the throat as deeply as possible.
– Gargling solution should be warm, as cold water will only worsen the condition, and hot water can cause burns and unnecessary pain and stress for the patient.
– Make the sound “ahhh” to improve tonsils irrigation and prevent interference of the tongue root with the passage of the solution.
– An important condition for proper gargling is the time of the procedure, at least 30 seconds is necessary for each gargling so that the liquid could thoroughly rinse the throat.
– In order not to swallow the solution, it is important to control the breathing.
OVERDOSE
The cases of overdose were not reported.
STORAGE CONDITIONS AND EXPIRATION DATE
The medicine should be stored out of reach of children, protected from light, at a temperature from + 2° C to + 25°C.
Expiration date: 24 months.
The information presented on the site should not be used for self-diagnosis and treatment and can not serve as a substitute for a doctor's internal consultation.
DENTOVITAL
- Without prescription
- Download instruction
- Share this:
- ABOUT PRODUCT
- INSTRUCTIONS
COMPOSITION AND PACKING
Syrup of herbal extracts, brown viscous liquid with individual odor (may have light sediment and clouding) in the original vials of 150 ml with a protective seal – (1) cardboard packs with protective branded holographic label; (2) Spoon – dispenser; (3) instruction for use.
Composition:
Common Sage Extract (Salvia officinalis L.)
Licorice Extract (Glycyrrhiza glabra L.)
Milfoil Extract (Achillea millefolium L.)
Chamomile Extract (Matricaria)
Marigold Extract (Calendula officinalis)
EFFECT OF THE MEDICINE
– Antimicrobial and anti-inflammatory action;
– Astriction and antiseptic action;
– Local anesthetic.
SIDE EFFECTS
There may be rarely allergic hypersensitivity to separate ingredients of the preparation.
Use of the medicine should be discontinued when hypersensitivity reactions occur.
SPECIAL INSTRUCTIONS
Before use, carefully shake the vial.
Keep the vial in a cool place after opening it. Do not heat and do not freeze.
INTERACTION WITH OTHER MEDICATION
When prescribing with other medication adverse effects were not revealed.
indications
Dentovital is used in the treatment and prevention of infectious and inflammatory diseases of the oral cavity:
– Stomatitis: acute, ulcerative, bacterial, ulcerous-erosive;
– Periodontitis: slight, moderate, severe;
– Gingivitis: acute, chronic, necrotizing ulcerative, atrophic;
– Flux (odontogenic periostitis). Scurvy (in the complex therapy);
– Hygiene treatment of removable dentures;
– To eliminate bad breath.
CONTRAINDICATIONS
Individual intolerance to certain ingredients of the medicine.
DOSAGE
The duration of treatment and the dose are determined by the attending physician for each patient individually.
For mouthwash, dilute 10-15 ml of the medicine with 100 g of water at a room temperature.
In acute diseases, the dose can be increased up to 30 ml.
It is recommended to rinse the mouth 3-4 times a day or after every meal.
The course of administration is 7 days.
OVERDOSE
The cases of overdose were not reported.
STORAGE CONDITIONS AND EXPIRATION DATE
The medicine should be stored out of reach of children, protected from light, at a temperature from + 2° C to + 25°C.
Expiration date: 24 months.
The information presented on the site should not be used for self-diagnosis and treatment and can not serve as a substitute for a doctor's internal consultation.
KASHLINET
- Without prescription
- Download instruction
- Share this:
- ABOUT PRODUCT
- INSTRUCTIONS
COMPOSITION AND PACKING
Syrup of herbal extracts, brown viscous liquid with individual odor (may have light sediment and clouding) in the original vials of 150 ml with a protective seal – (1) cardboard packs with protective branded holographic label; – (2) Spoon – dispenser; (3) instruction for use.
Composition:
Athaea Extract (Althaea officinalis)
Camel’s-thorn Extract (Alhagi)
Chamomile Extract (Matricaria chamomilla)
Licorice Extract (Glycyrrhiza glabra)
EFFECT OF THE MEDICINE
– Expectorant;
– Anti-inflammatory;
– Effect on the mucous membranes of the respiratory tract, thins mucus and reduces its viscosity.
SIDE EFFECTS
There may be rarely allergic hypersensitivity to separate ingredients of the preparation.
Use of the medicine should be discontinued when hypersensitivity reactions occur.
SPECIAL INSTRUCTIONS
Before use, carefully shake the vial.
Keep the vial in a cool place after opening it. Do not heat and do not freeze.
INTERACTION WITH OTHER MEDICATION
When prescribing with other medication adverse effects were not revealed.
INDICATIONS
KASHLINET is indicated for acute and chronic inflammatory diseases which are accompanied by difficult coughing with phlegm:
– Tracheitis;
– Bronchitis;
– Pneumonia;
– Bronchiectasis;
– Pulmonary emphysema (in adults and children aged 1-year-old and above, in such cases KASHLINET helps to improve and facilitate the expectoration of sputum).
CONTRAINDICATIONS
Individual intolerance to certain ingredients of the medicine.
DOSAGE
For peroral use.
The duration of treatment and the dose are determined by the attending physician for each patient individually.
CHILDREN BETWEEN 1 AND 6 YEARS OF AGE
CHILDREN BETWEEN 7 AND 12 YEARS OF AGE
ADULTS AND CHILDREN ABOVE 12 YEARS OF AGE
2.5 ml (1/2 teaspoon) 3 times a day
2.5-5.0 ml (1/2-1 teaspoon) 3 times a day
5.0-7.5 ml (2-3 teaspoon) 3 times a day
The medicine is administered regardless of meals.
The course of administration is usually from 3 to 7 days.
OVERDOSE
The cases of overdose were not reported.
STORAGE CONDITIONS AND EXPIRATION DATE
The medicine should be stored out of reach of children, protected from light, at a temperature from + 2° C to + 25°C.
Expiration date: 24 months.
The information presented on the site should not be used for self-diagnosis and treatment and can not serve as a substitute for a doctor's internal consultation.
BRONKHODOKS
- Without prescription
- Download instruction
- Share this:
- ABOUT PRODUCT
- INSTRUCTIONS
COMPOSITION AND PACKING
Syrup of herbal extracts, brown viscous liquid with individual odor (may have light sediment and clouding) in the original vials of 150 ml with a protective seal – (1) cardboard packs with protective branded holographic label; (2) spoon – dispenser; (3) instruction for use.
|
|
||
| Common Sage Extract (Salvia officinalis L.) | ||
| Coltsfoot Extract (Tussilago L) | ||
| Licorice Extract (Glycyrrhiza glabra L.) | ||
| Aloe Extract (Aloe arborescens Mill.) | ||
| Plantain Extract (Plantago L) |
EFFECT OF THE MEDICINE
Bronkhodoks is a combination medicine with antitussive and bronchodilatory action.
– It has a suppressive effect on the cough center without affecting breathing.
– It also has weak bronchospasmolytic and secretolytic action.
– The medicine stimulates respiration, expands bronchi and reduces bronchial mucosa edema.
– It has anti-inflammatory and weak antimicrobial effect.
INDICATIONS
Bronkhodoks is used in combination therapy for the treatment of the followings:
- Acute and chronic bronchitis;
- Tracheobronchitis;
- Bronchial asthma;
- Bronchiectasis;
- Sanitation of a bronchial tree in pre – and postoperative period;
- Atelectasis when mucous plug blocks bronchi;
- Pneumonia;
- Pertussis;
- Tuberculosis.
- To eliminate the symptoms of cough and bronchoconstriction
SPECIAL INSTRUCTIONS
Before use, carefully shake the vial.
Keep the vial in a cool place after opening it. Do not heat and do not freeze.
INTERACTION WITH OTHER MEDICATION
When prescribing with other medication adverse effects were not revealed.
CONTRAINDICATIONS
Individual intolerance to certain ingredients of the medicine.
Hyperacidity gastritis. Pregnancy. Lactation.
DOSAGE
The duration of treatment and the dose are determined by the attending physician for each patient individually.
ADULTS AND CHILDREN ABOVE 12 YEARS OF AGE
CHILDREN BETWEEN 3 AND 12 YEARS OF AGE
5.0-7.5 ml, 3 times a day
2.5-5.0 ml, 3 times a day
For peroral use, 10-15 minutes after the meal.
The course of administration is 14 days
There may be rarely allergic hypersensitivity to separate ingredients of the preparation.
Use of the medicine should be discontinued when hypersensitivity reactions occur.
OVERDOSE
The cases of overdose were not reported.
STORAGE CONDITIONS AND EXPIRATION DATE
The medicine should be stored out of reach of children, protected from light, at a temperature from + 2° C to + 25°C.
Expiration date: 24 months.
The information presented on the site should not be used for self-diagnosis and treatment and can not serve as a substitute for a doctor's internal consultation.